Number of pages: 100 | Report Format: PDF | Published date: May 31, 2023
Historical Years – 2021 | Base Year – 2022 | Forecasted Years – 2023-2031
|
Report Attribute |
Details |
|
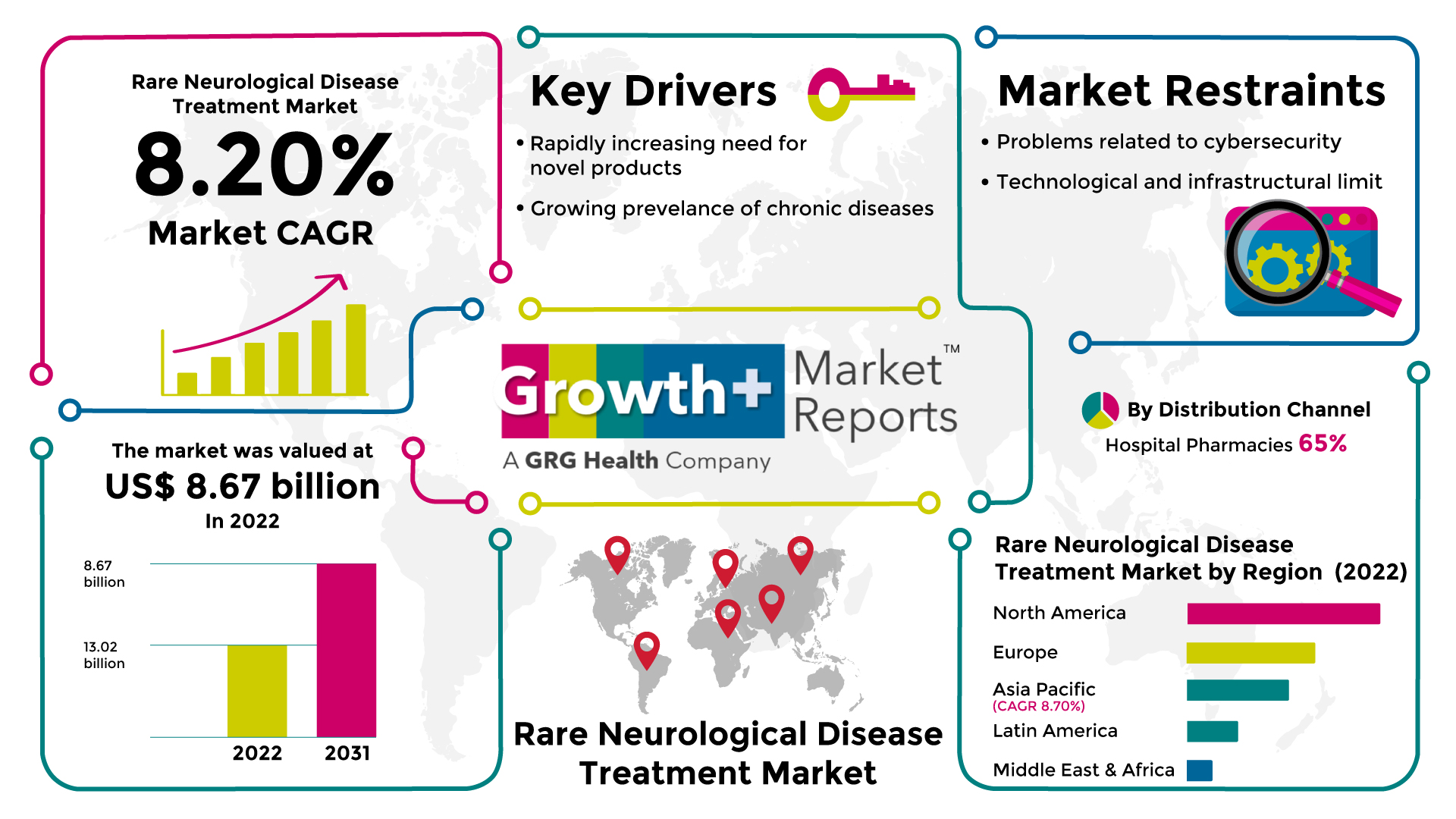
Market Size Value in 2022 |
US$ 8.67 billion |
|
Revenue Forecast in 2031 |
US$ 13.02 billion |
|
CAGR |
8.2% |
|
Base Year for Estimation |
2022 |
|
Forecast Period |
2023 to 2031 |
|
Historical Year |
2021 |
|
Segments Covered |
Drug Type, Indication, Distribution Channel, and Region |
|
Regional Scope |
North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
According to the deep-dive market assessment study by Growth Plus Reports, the rare neurological disease treatment market was valued at US$ 8.67 billion in 2022 and is expected to register a revenue CAGR of 8.2% to reach US$ 13.02 billion by 2031.
Rare Neurological Disease Treatment Market Fundamentals
A rare neurological disease is a neurological disorder that affects a relatively small number of people compared to more common neurological conditions. These diseases often have low prevalence rates, meaning they are uncommon in the general population. Less than 1 in 100,000 individuals are affected by rare neurological illnesses. Because it can enhance the quality of life for those who suffer from these disorders, treatment for rare neurological diseases is crucial. Pharmaceutical companies research and develop medications specifically targeted at managing symptoms or slowing the progression of rare neurological disorders. These treatments may involve the use of drugs, such as disease-modifying therapies, immunomodulators, or symptom-management medications. Gene therapy holds significant potential for treating rare neurological diseases caused by genetic mutations. It involves introducing genetic material into a patient's cells to correct or replace the faulty genes responsible for the condition.
Rare Neurological Disease Treatment Market Dynamics
The global rare neurological disease treatment market has significant growth potential due to several key drivers that are fueling advancements and investment in this field. In terms of disease diagnosis, treatment, and management, uncommon neurological diseases place a major burden on healthcare systems. Narcolepsy, amyotrophic lateral sclerosis, Alzheimer's disease, multiple sclerosis, spinal muscular atrophy (SMA), Duchenne muscular dystrophy, and others are among the most common uncommon neurological illnesses. 7,000 uncommon neurological disorders have been documented in the U.S., according to statistics from the National Institute of Neurological Disorders and Stroke published in 2019. Additionally, a large amount of morbidity and mortality are caused by these disorders, according to the same study. The major causes of an increase in the prevalence of rare neurological illnesses include sleep deprivation, psychological stress, an inherited genetic defect, abrupt changes in sleep patterns, and aging. These variables make the elderly more susceptible to neurological ailments.
Additionally, these illnesses increase morbidity and disability, which impacts the country's economy. As the world's population ages, there will likely be an increase in neurological sleep-related illnesses in the years to come. Around 40 million Americans suffer from neurological illnesses, and another 20 million occasionally have sleeping issues, according to data from the National Institutes of Health (NIH) published in 2019. Precision medicine, which focuses on tailoring treatments to an individual's specific genetic, molecular, and clinical characteristics, has revolutionized healthcare. The understanding of rare neurological diseases at a molecular level has increased, enabling the development of targeted therapies. Advances in genomics, proteomics, and biomarker identification have paved the way for personalized treatment approaches that address the underlying causes of these diseases. These growth drivers provide a favorable environment for research, development, and investment in the treatment market for rare neurological diseases.
However, rare neurological diseases affect a small number of individuals. Due to the potential lack of profitability, the limited patient population makes it financially challenging for pharmaceutical companies to invest in research and treatment development. This limitation often leads to a scarcity of funding and resources for developing therapies for these diseases. The market for treatments for rare neurological diseases is also anticipated to grow slowly due to the high cost of those treatments.
Rare Neurological Disease Treatment Market Ecosystem
The global rare neurological disease treatment market is analyzed from the following perspectives: drug type, indication, distribution channel, and region.
Rare Neurological Disease Treatment Market by Drug Type
Based on the drug type, the global rare neurological disease treatment market is segmented into biologics and small molecules.
Small molecules account for the largest revenue share in the global rare neurological disease treatment market. A small molecule is a drug that can enter cells quickly due to its low molecular weight. The segment is dominating due to the higher cost of biological drugs. Small molecules can be utilized to treat genetic epilepsies, including uncommon or ultrarare genetic epilepsy, according to a study report published in August 2021 in the Neurotherapeutics Journal. Small compounds have the benefit of being able to access extracellular and intracellular targets, allowing for the control of specific protein activities like ion channel gating. Additionally, these chemicals can be produced more rapidly, in greater quantities, and at a lower cost, making them advantageous for the future development of medications for uncommon diseases. Such investigations will likely open up new possibilities and boost demand for small molecules in treating uncommon neurological illnesses.
Rare Neurological Disease Treatment Market by Indication
Based on the indication, the global rare neurological disease treatment market is segmented into narcolepsy, Creutzfeldt-Jakob disease, multiple sclerosis, spinal muscular atrophy, and others.
Creutzfeldt-Jakob disease (CJD) segment held a significant revenue share in the global rare neurological disease treatment market. CJD is a rare and fatal degenerative neurological disorder. CJD is the most common form, accounting for about 85% of cases. The estimated annual incidence of CJD varies geographically, ranging from about 0.5 to 1.5 cases per million population.
Rare Neurological Disease Treatment Market by Distribution Channel
Based on the distribution channel, the global rare neurological disease treatment market is segmented into hospital pharmacies, retail pharmacies, and online pharmacies.
Hospital pharmacies account for the largest revenue share of over 65% of the global rare neurological disease treatment market. This is because hospitals have access to many different medications for treating neurological problems. Additionally, hospital-based pharmaceutical firms are working on research projects to create new treatments that will be less likely to cause adverse effects than previous medications. Due to expanding public awareness of health and safety issues and an increase in the prevalence of rare neurological diseases like Parkinson's disease and Alzheimer's disease worldwide, retail pharmacies are predicted to have the fastest growth during the forecast period.
Rare Neurological Disease Treatment Market by Region
Geographically, the global rare neurological disease treatment market has been segmented into North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
North America accounts for a significant revenue share in the rare neurological disease treatment market. This is because reimbursement is available for treating uncommon diseases, the prevalence of rare neurological disorders is rising, and regional research and development (R&D) is growing. According to a StatPearls article updated in March 2022, about 350 instances of Creutzfeldt-Jakob disease (CJD) are diagnosed each year in the United States, which is consistent with an Orphanet report released in January 2022. The most prevalent type of human prion disease is sporadic CJD, with a mean age of onset of 61 years. Therefore, throughout the forecast period, the high prevalence in the region is anticipated to fuel demand for the market for treating uncommon neurological diseases.
Additionally, the strategic actions taken by market participants, such as product launches, approvals, collaborations, and partnerships, support the expansion of the market under investigation. For instance, the US FDA granted Novartis Fast Track status in December 2021 for branaplam (LMI070), a medication used to treat HD. During the forecast period, product launches and approvals are anticipated to fuel the market in the area. Rising healthcare R&D spending and the availability of established healthcare infrastructure are other major contributors to the regional market's growth. For instance, according to the National Institutes of Health (NIH) RePORT 2022 report, the amount spent on research into Huntington's disease in 2020 and 2021 was 49 and 46 million dollars, respectively. In the United States, R&D expenditures on Huntington's disease are projected to total USD 48 million in 2022.
Due to the large unmet clinical needs, quickly developing healthcare infrastructure, and economic development, particularly in India, China, Japan, Singapore, and South Korea, Asia Pacific is anticipated to grow at the quickest rate throughout the projection period.
Rare Neurological Disease Treatment Market Competitive Landscape
The prominent players operating in the global rare neurological disease treatment marketare: -
Rare Neurological Disease Treatment Market Strategic Developments
A rare neurological disease is a neurological disorder that affects a relatively small number of people compared to more common neurological conditions.
The global rare neurological disease treatment market during the forecast period is expected to grow at a revenue CAGR of 8.2%.
Bayer AG, Pfizer Inc., Novartis AG, Merck & Co. Inc., and CSL Ltd. are the major players in the rare neurological disease treatment market.
The Asia Pacific region is projected to grow at a high revenue CAGR in the global rare neurological disease treatment market.
Due to the high costs associated with developing treatments for rare diseases, the reimbursement landscape can present challenges.
*Insights on financial performance are subject to the availability of information in the public domain